 Thus, on moving dwon a group, the atomic radii of elements increase gradually. In general, anions are larger than the corresponding neutral atom, since adding electrons increases the number of electron-electron repulsion interactions that take place. In a group, the distance between the valence electron and nucleus increases with increase in nuclear charge. The following charts illustrate the general trends in the radii of atoms: The sizes of cations and anions follow similar trends to those of neutral atoms. As a result the distance between the nucleus and the valence shell increases. On moving dwon a group, a new shell of electrons is added. Variation of atomic radii (atomic size) in a group: Thus, on moving from left to right in a period, there is a trend of decrease in atomic radii. Due to this increased nuclear charge, the attraction between electron and nucleus increases, hence the atomic size decreases. In a period in the periodic table, the nuclear charge increases, and the new. We can explain this trend by considering the nuclear charge and energy level. 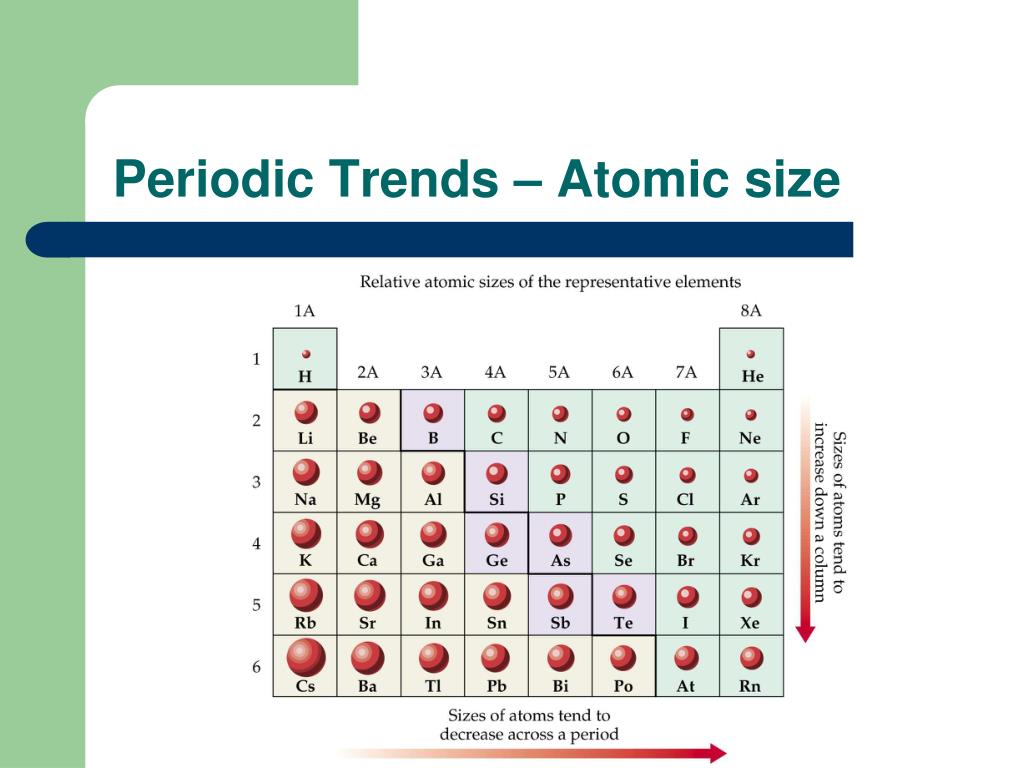
On moving from left to right in a period, the atomic number of element increases successively by 1. Consequently, from top to bottom in a group, the size of the atom increases. The atomic radii of elements vary in the periodic table in a fixed pattern. Because of these two trends, the largest atoms are found in the lower left corner of the periodic table, and the smallest are found in the upper right corner ( Figure 4.3.4 ). atomic radius.Ītomic radius : The average distance between the centre of the nucleus and the outermost shell which contains electrons in an isolated atom is called atomic radius.Ītomic radius may be expressed in angstroms ( A 0 ) or centimeter (cm) or in picometers (pm). In the periodic table, atomic radii decrease from left to right across a row and increase from top to bottom down a column. If an atom is considered to be a sphere, then the term. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
